The method is based on extraction from the filtrates of water-plant systems by means of solvents. Generally, caffeine is extracted from plants such as cacao, tea and coffee. Overall, caffeine is found in the beans, leaves, and fruit of over 60 plants, where it acts as a natural pesticide that paralyzes and kills certain insects feeding upon them. It is found in the leaves and beans of the coffee plant, in tea, yerba mate, and guarana berries, and in small quantities in cocoa, the kola nut and the Yaupon Holly. Caffeine is the world's most widely consumed psychoactive substance. Beverages containing caffeine, such as coffee, tea, soft drinks and energy drinks enjoy great popularity. Early investigators (Gürü and İçen, 2004) indicate that caffeine is a central nervous system (CNS) stimulant, having the effect of temporarily warding off drowsiness, restoring alertness and muscle relaxant properties. As with all methylxanthines, caffeine has low solubility and is therefore often combined with a wide variety of compounds to form complexes, such as the double salt sodium benzoate, for purposes of enhanced solubility in consumer goods like soft drinks. In its pure form, caffeine occurs as odorless, white, fleecy masses, glistening needles or powder. It belongs to the group of chemicals known as methylxanthines, which also includes the closely related chemicals theophylline and theobromine. Correlation.Ĭaffeine is an alkaloid, or nitrogen-containing substance, with the chemical formula C 8H 10N 4O 2. The solubility of caffeine in chloroform and dichloromethane was high compared with other solvents 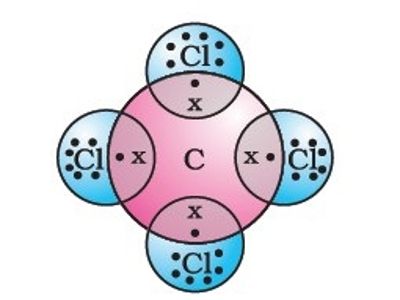
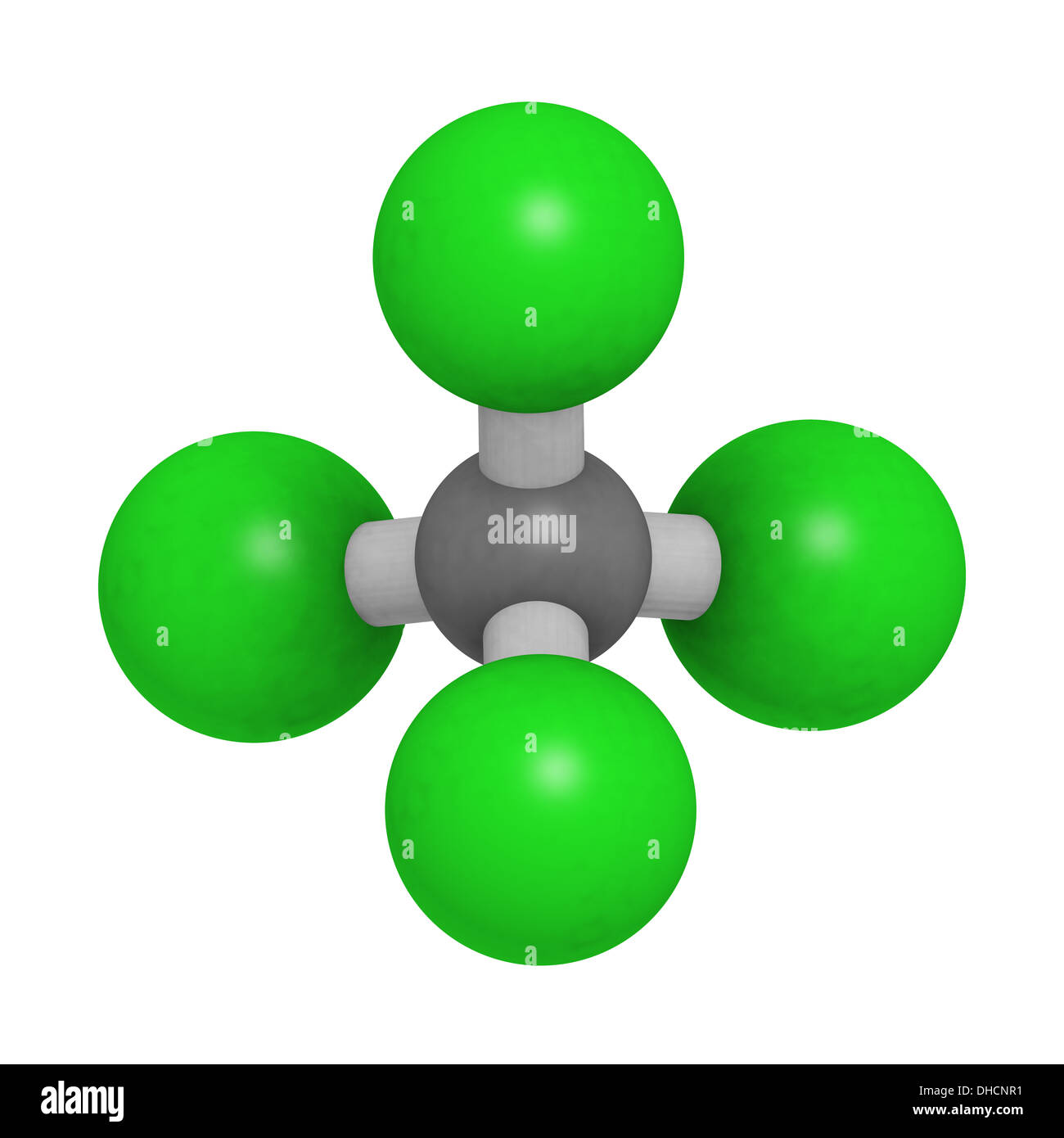
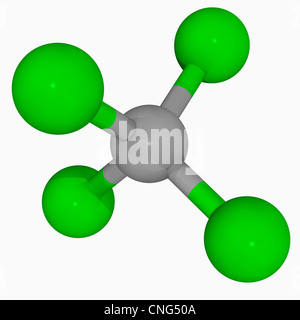
The assessment is scheduled to be finalized by June.Solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane, and acetone between 298 and 323 KĬhemical Industries Department, Iranian Research Organization for Science and Technology (IROST), Tehran, - The solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane and acetone were measured by a gravimetrical method from (298 to 323) K and the solubility data was correlated against temperature. In a recent draft risk assessment of CCl 4 standards under the Toxic Substances Control Act, the US Environmental Protection Agency reports that the compound is safe to use in manufacturing plants if workers wear appropriate personal protective equipment. The CCl 4 currently produced is used as a feedstock for ozone-safe refrigerants and in some minor agricultural and industrial processes. What little is available today is a coproduct from the manufacture of chloroform and dichloromethane however, US production is still 15 times the amount called for in the Montreal Protocol. In its heyday, CCl 4 was produced by chlorinating C 1 compounds such as chloroform, carbon disulfide, and, in later years, methane. Consequently, production and use have been significantly reduced. CCl 4 is also a “greenhouse gas” that, according to the 1995 Montreal Protocol, depletes ozone in the Earth’s stratosphere. It was also a widely used dry-cleaning solvent, refrigerant, and fire suppressant.ĭuring the 1970s and ’80s, however, toxicologists discovered that inhalation of or skin contact with carbon tetrachloride (CCl 4) can damage many organs, including the liver, kidneys, and central nervous system (see hazard table). Readers of a certain age can remember when “carbon tet” was the go-to household solvent that easily removed organic residues such as adhesives and oils from almost any surface.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
